Manganese has a bittersweet astringent taste and is detectable at concentrations above 0.05 ppm by many people.
Manganese is often found with iron, and causes brown, black and/or tea-colored staining, and imparts asphaltic and oily tastes, above .05 ppm. Some of the same filters used for iron, are used for manganese removal.
Iron and manganese are often found in a dissolved state in well waters, and the water appears clear when first drawn. Upon exposure to air, or after the addition of oxidants (such as chlorine bleach or ozone), this ferrous iron is oxidized (“rusted”) to the ferric state to form insoluble particles.
The water then looks orange or yellow, or in the case with manganese, brown or black. This can happen in toilet flush tanks and in the washing machine or dishwasher.
If sulfur odor is present along with iron, typically chlorine or ozone is fed ahead of an iron filter.
There are four main types of iron or conditions in which iron is found in well water:
- Soluble “Clear Water” Iron
- Insoluble Oxidized “Red Water” Iron
- Organic Iron
- Iron Bacteria
Questions to Ask When Choosing an Iron Filter:
1. What type of iron do I have in my water system?
2. What is the flow rate I have available, to backwash the iron filter I choose?
3. Do I have manganese, hydrogen sulfide odor, iron bacteria or tannin in my water?
4. What is the pH (acid or alkaline nature) of my water?
5. According to the water test results, will the water treatment unit remove the total iron concentration?
If there is an iron problem with the water supply, the first step is to determine the source. The source of iron may be from the corrosion of iron or steel pipes or other components of the plumbing system where the acidity of the water, measured as pH, is below 6.8.
A water test to determine your water's iron, pH, manganese, hardness and total dissolved solids is a good place to begin. If the water direct from the well has a yellow or tea colored look to it, an additional test for tannin is recommended as well. Take the sample as close to the well as possible.
With these results, you can identify if you need any type of water treatment, and what type of system to select, based on your water chemistry.
If the source of water is a public water system and you experience iron-related problems, it is important to contact a utility official to determine whether the red water is from the public system or from the home's plumbing or piping.
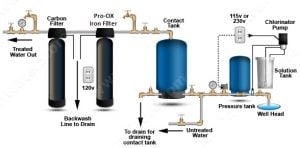
Our main recommendations for iron treatment is the long-lasting Pro-OX Iron Filtration or the Pro-OX-AIR if odors are present.
For water very high in iron with high levels of hydrogen sulfide and/or manganese, and/or iron bacteria, we recommend a pre-chlorination feed for maximum removal rates.
After the iron filter, many folks will use a carbon filter to remove any trace of the chlorine taste or odor.
The role of pH is very important in iron treatment. Generally, if the pH of the water is acidic (or less than 7.0), it must be corrected with a special type of neutralizer filter ahead of the iron filtration system. It is usually best to test for pH right at the water source, and not depend on laboratory analysis for pH, since in some cases the pH can raise after sampling, giving false results.
Water softening is sometimes used to remove dissolved iron in low levels (less than 2.0 ppm), although more commonly, we use iron filters in conjunction with water softening if the water is both hard, and high in iron or manganese. Iron is generally not thought of as toxic, although there are some studies that show liver toxicity to men.
Need help selecting a system? Fill out our quick and easy Water System Designer Form and get a response in 24 hours or less.
Typical Chlorine Injection System with Pro-OX Iron Filter and Carbon Backwash Filter. Ultimate system removes iron, manganese, odor, kills iron bacteria. No chlorine residual in the house. No chlorine tastes or odors.